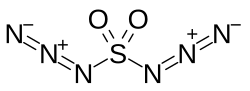
Sulfuryl diazide
 | |
| Names | |
|---|---|
| IUPAC name
Sulfuryl diazide
| |
| Other names
Sulfuryl azide; Sulfonyl diazide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value). Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value).Lua error in Module:EditAtWikidata at line 29: attempt to index field 'wikibase' (a nil value). |
| E number | Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value). |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| SO2(N3)2 | |
| Molar mass | 148.10 g·mol−1 |
| Melting point | −15 °C (5 °F; 258 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sulfuryl diazide or sulfuryl azide is a chemical compound with the molecular formula SO2(N3)2. It was first described in the 1920s when its reactions with benzene and p-xylene were studied by Theodor Curtius and Karl Friedrich Schmidt.[1][2][3] The compound is reported as having "exceedingly explosive, unpredictable properties" and "in many cases very violent explosions occurred without any apparent reason".[1]
Synthesis
[edit | edit source]It was not until 2011 that sulfuryl diazide was isolated in a pure enough state to be fully characterized.[4] It was characterized by infrared and Raman spectroscopy; its structure in the solid state was determined by x-ray crystallography.[4] Its melting point is -15 °C.[4] It was prepared by the reaction of sulfuryl chloride (SO2Cl2) with sodium azide (NaN3) using acetonitrile as solvent:
- SO2Cl2 + 2 NaN3 → SO2(N3)2 + 2 NaCl
Physical properties
[edit | edit source]Sulfuryl diazide is extremely explosive and can explode even without strong external influences. The compound crystallizes in the monoclinic crystal system in the space group C2/c (space group no. 15) with the lattice parameters a = 24.3405 Å; b = 5.41599 Å , c = 17.2915 Å and β = 111.819°.[5] Photolysis in an argon matrix initially yields a short-lived nitrene. Further decomposition leads primarily to elemental nitrogen and sulfur dioxide.[6]
Uses
[edit | edit source]Sulfuryl diazide has been used as a reagent to perform reactions that remove nitrogen from heterocyclic compounds:[7][8][9]
- R1−NH−R2 + SO2(N3)2 → R1−R2 + SO2 + 2 N2 + HN3
See also
[edit | edit source]References
[edit | edit source]- ^ a b Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ a b c Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).