Pyramidal inversion

|
⇌ | 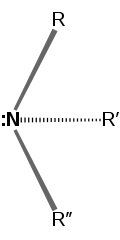
|
| Nitrogen inversion: The amine C3 axis is horizontal; the pair of dots represent the lone pair (on that axis). Note that the two amine molecules are symmetric across a mirror plane. If the three R groups attached are all unique, then the amine is chiral; isolability depends on the free energy required to invert the molecule. | ||
In chemistry, pyramidal inversion (also umbrella inversion) is a fluxional process in compounds with a pyramidal molecule, such as ammonia (NH3) "turns inside out".[1][2] It is a rapid oscillation of the atom and substituents, the molecule or ion passing through a planar transition state.[3] For a compound that would otherwise be chiral due to a stereocenter, pyramidal inversion allows its enantiomers to racemize. The general phenomenon of pyramidal inversion applies to many types of molecules, including carbanions, amines, phosphines, arsines, stibines, and sulfoxides.[4][2]
Energy barrier
[edit | edit source]
The identity of the inverting atom has a dominating influence on the barrier. Inversion of ammonia is rapid at room temperature, inverting 30 billion times per second. Three factors contribute to the rapidity of the inversion: a low energy barrier (24.2 kJ/mol; 5.8 kcal/mol), a narrow barrier width (distance between geometries), and the low mass of hydrogen atoms, which combine to give a further 80-fold rate enhancement due to quantum tunnelling.[5] In contrast, phosphine (PH3) inverts very slowly at room temperature (energy barrier: 132 kJ/mol).[6] Consequently, amines of the type RR′R"N usually are not optically stable (enantiomers racemize rapidly at room temperature), but P-chiral phosphines are.[7] Appropriately substituted sulfonium salts, sulfoxides, arsines, etc. are also optically stable near room temperature. Steric effects can also influence the barrier.
Nitrogen inversion
[edit | edit source]
Pyramidal inversion in nitrogen and amines is known as nitrogen inversion.[8] It is a rapid oscillation of the nitrogen atom and substituents, the nitrogen "moving" through the plane formed by the substituents (although the substituents also move - in the other direction);[9] the molecule passing through a planar transition state.[10] For a compound that would otherwise be chiral due to a nitrogen stereocenter, nitrogen inversion provides a low energy pathway for racemization, usually making chiral resolution impossible.[11]
Quantum effects
[edit | edit source]Ammonia exhibits a quantum tunnelling due to a narrow tunneling barrier,[12] and not due to thermal excitation. Superposition of two states leads to energy level splitting, which is used in ammonia masers.
Examples
[edit | edit source]The inversion of ammonia was first detected by microwave spectroscopy in 1934.[13]
In one study the inversion in an aziridine was slowed by a factor of 50 by placing the nitrogen atom in the vicinity of a phenolic alcohol group compared to the oxidized hydroquinone.[14]

The system interconverts by oxidation by oxygen and reduction by sodium dithionite.
Exceptions
[edit | edit source]
Conformational strain and structural rigidity can effectively prevent the inversion of amine groups. Tröger's base analogs[16] (including the Hünlich's base[17]) are examples of compounds whose nitrogen atoms are chirally stable stereocenters and therefore have significant optical activity.[15]
References
[edit | edit source]- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ a b IUPAC, Compendium of Chemical Terminology, 5th ed. (the "Gold Book") (2025). Online version: (2006–) "Pyramidal inversion". Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Control of Pyramidal Inversion Rates by Redox Switching Mark W. Davies, Michael Shipman, James H. R. Tucker, and Tiffany R. Walsh J. Am. Chem. Soc.; 2006; 128(44) pp. 14260–14261; (Communication) Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ a b Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).