Oxidative coupling
Oxidative coupling in chemistry is a coupling reaction of two molecular entities through an oxidative process. Usually oxidative couplings are catalysed by a transition metal complex like in classical cross-coupling reactions, although the underlying mechanism is different due to the oxidation process that requires an external (or internal) oxidant.[1][2] Many such couplings utilize dioxygen as the stoichiometric oxidant but proceed by electron transfer.[3]
C-C Couplings
[edit | edit source]Many oxidative couplings generate new C-C bonds. Early examples involve coupling of terminal alkynes:[4]
- 2 RC≡CH + 2 Cu(I) → RC≡C-C≡CR + 2 Cu + 2 H+
Aromatic coupling
[edit | edit source]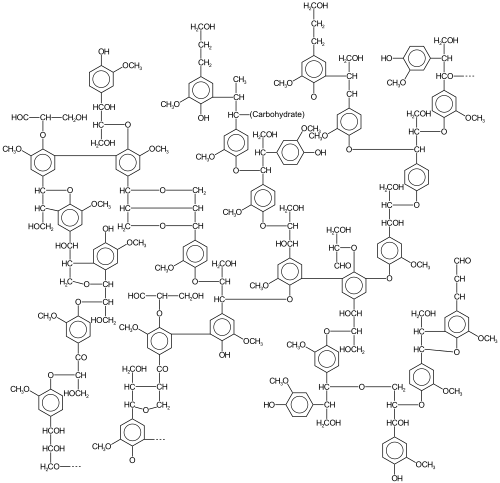
In oxidative aromatic coupling the reactants are electron-rich aromatic compounds. Typical substrates are phenols and typical catalysts are copper and iron compounds and enzymes,[6] although Scholl demonstrated that high heat and a Lewis acid suffice. The first reported synthetic application dates back to 1868 with Julius Löwe and the synthesis of ellagic acid by heating gallic acid with arsenic acid or silver oxide.[7] Another reaction is the synthesis of 1,1'-Bi-2-naphthol from 2-naphthol by iron chloride, discovered in 1873 by Alexander Dianin[8] (S)-BINOL can be prepared directly from an asymmetric oxidative coupling of 2-naphthol with copper(II) chloride.[9]

Coupling of methane
[edit | edit source]Coupling reactions involving methane are highly sought, related to C1 chemistry because C2 derivatives are far more valuable than methane. The oxidative coupling of methane gives ethylene:[10][11]
- 2CH
4 + O
2 → C
2H
4 + 2H
2O
Other oxidative couplings
[edit | edit source]
The oxygen evolution reaction entails, in effect, the oxidative coupling of water molecules to give O2.
References
[edit | edit source]- ^ Oxidative Cross-Coupling Reactions. Aiwen Lei, Wei Shi, Chao Liu, Wei Liu, Hua Zhang, Chuan He, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany (2017). Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ IUPAC. Compendium of Chemical Terminology, 2nd ed. (the "Gold Book"). Compiled by A. D. McNaught and A. Wilkinson. Blackwell Scientific Publications, Oxford (1997). Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Grzybowski, M., Skonieczny, K., Butenschön, H. and Gryko, D. T. (2013), Comparison of Oxidative Aromatic Coupling and the Scholl Reaction Angew. Chem. Int. Ed., 52: 9900–9930. Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Löwe, Zeitschrift für Chemie, 1868, 4, 603
- ^ A. P. Dianin, Zh. Russ. Fiz.-Khim. O-va. 1874, 183
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value).
- ^ Olah, G., Molnar, A. "Hydrocarbon Chemistry" John Wiley & Sons, New York, 2003. Lua error in Module:Citation/CS1/Configuration at line 2172: attempt to index field '?' (a nil value)..