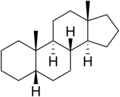
Androstane
Jump to navigation
Jump to search
 | |
| Names | |
|---|---|
| IUPAC name
5ξ-Androstane
| |
| Systematic IUPAC name
(3aS,3bS,5aΞ,9aS,9bS,11aS)-9a,11a-Dimethylhexadecahydro-1H-cyclopenta[a]phenanthrene | |
| Other names
Etioallocholane; 10β,13β-Dimethylgonane
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value). Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value).Lua error in Module:EditAtWikidata at line 29: attempt to index field 'wikibase' (a nil value). |
| E number | Lua error in Module:Wikidata at line 880: attempt to index field 'wikibase' (a nil value). |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C19H32 | |
| Molar mass | 260.465 g·mol−1 |
| Density | 0.95 g/ml |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Androstane is a C19 steroidal hydrocarbon with a gonane core. Androstane can exist as either of two isomers, known as 5α-androstane and 5β-androstane.
-
5α-Androstane
-
5β-Androstane
Pharmacology
[edit | edit source]5α-Androstane is reported to be effective as an androgen, in spite of having no oxygen containing functional groups.[1][2]
Androstanes
[edit | edit source]Androstanes are steroid derivatives with carbons present at positions 1 through 19.
See also
[edit | edit source]References
[edit | edit source]